You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
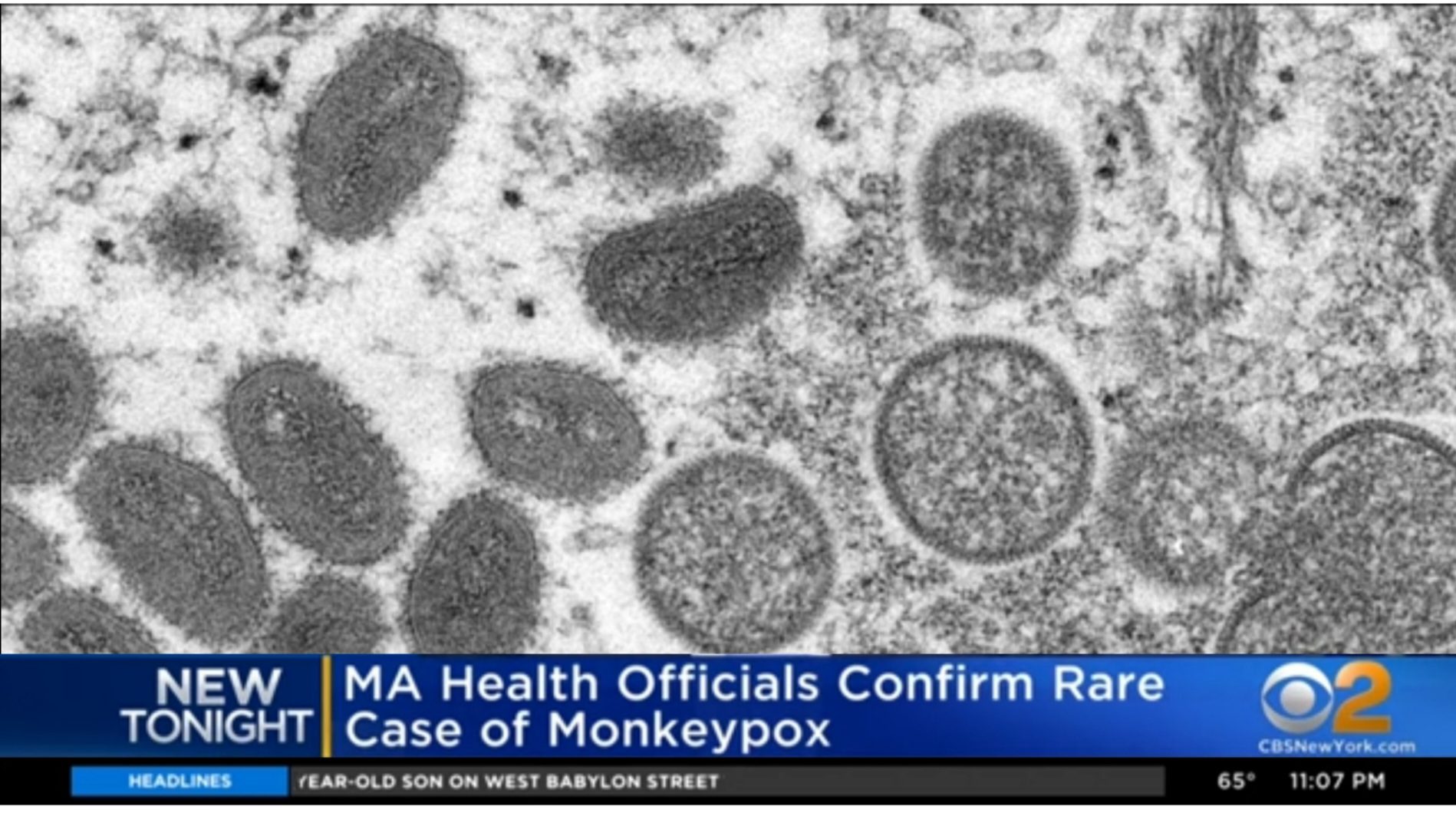
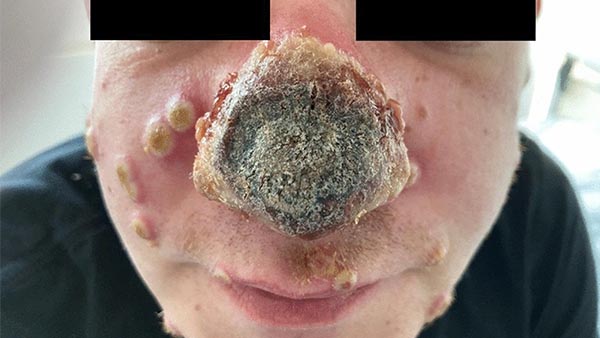
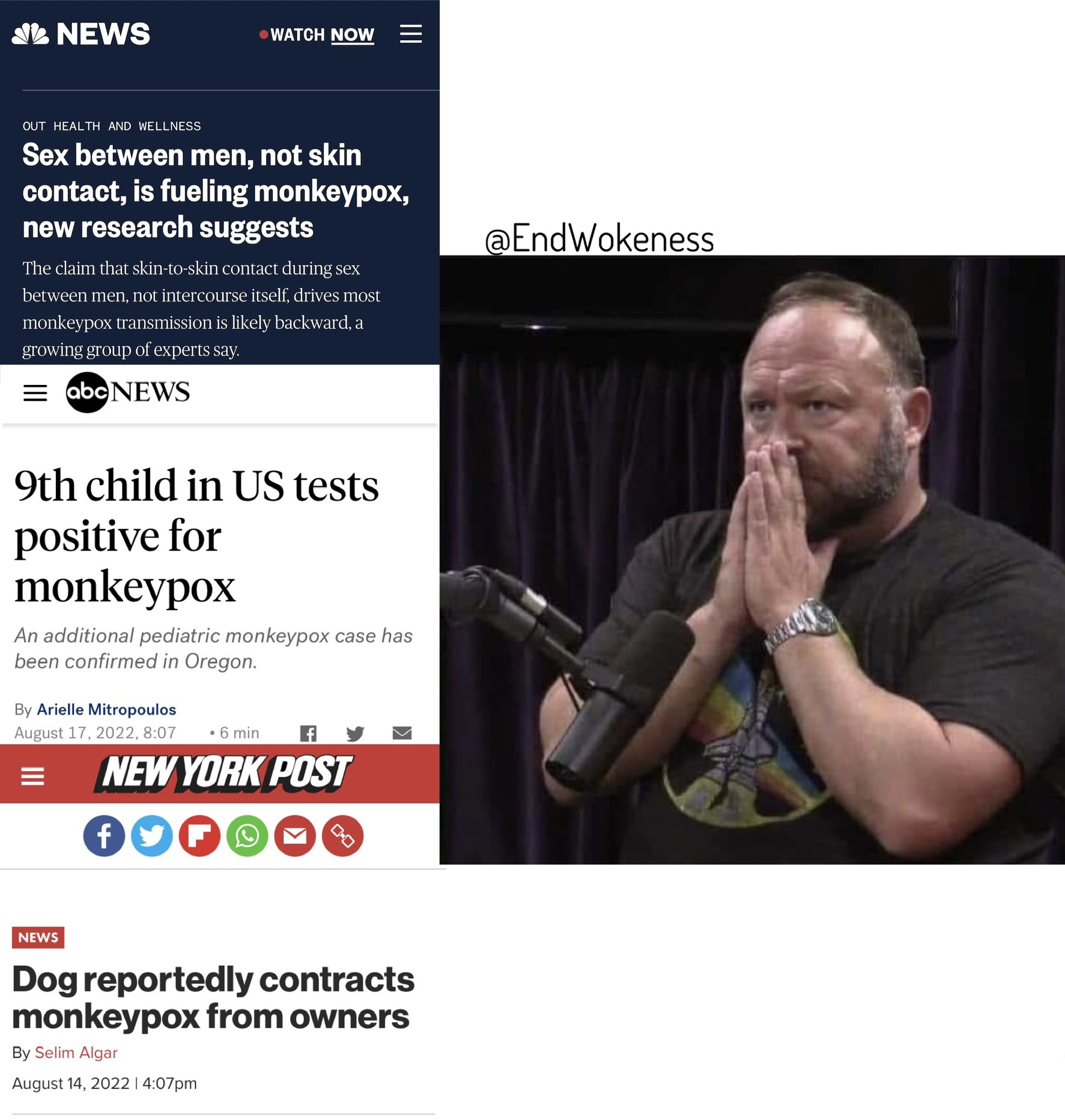
BREAK OUT the MONKEYPOX!!!
- Thread starter GasMan
- Start date
FDA emergency authorization. wait, where have I heard that before?  ... no no we're good because it is "safe and effective". my bad
... no no we're good because it is "safe and effective". my bad
Today, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for the JYNNEOS vaccine to allow healthcare providers to use the vaccine by intradermal injection [administered between layers of skin] for individuals 18 years of age and older who are determined to be at high risk for monkeypox infection.
JYNNEOS has been tested in individuals with immunocompromising conditions and has been found to be safe and effective in the trials that were performed to support approval. It was initially developed specifically as an alternative for use in immunocompromised individuals in the event of a smallpox outbreak.
On the basis of the determination by the Secretary of the Department of Health and Human Services on Aug. 9, 2022, that there is a public health emergency, or the significant potential for a public health emergency, that has a significant potential to affect national security or the health and security of United States citizens living abroad, and the declaration on Aug. 9, 2022, that circumstances exist justifying the emergency use of vaccines, the FDA may issue an EUA to allow emergency use of unapproved vaccines or unapproved uses of approved vaccines.
 ... no no we're good because it is "safe and effective". my bad
... no no we're good because it is "safe and effective". my badFDA Clears The Way For Monkeypox Vaccine
August 9, 2022Today, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for the JYNNEOS vaccine to allow healthcare providers to use the vaccine by intradermal injection [administered between layers of skin] for individuals 18 years of age and older who are determined to be at high risk for monkeypox infection.
JYNNEOS has been tested in individuals with immunocompromising conditions and has been found to be safe and effective in the trials that were performed to support approval. It was initially developed specifically as an alternative for use in immunocompromised individuals in the event of a smallpox outbreak.
On the basis of the determination by the Secretary of the Department of Health and Human Services on Aug. 9, 2022, that there is a public health emergency, or the significant potential for a public health emergency, that has a significant potential to affect national security or the health and security of United States citizens living abroad, and the declaration on Aug. 9, 2022, that circumstances exist justifying the emergency use of vaccines, the FDA may issue an EUA to allow emergency use of unapproved vaccines or unapproved uses of approved vaccines.
?FDA emergency authorization. wait, where have I heard that before?... no no we're good because it is "safe and effective". my bad
FDA Clears The Way For Monkeypox Vaccine
August 9, 2022
Today, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for the JYNNEOS vaccine to allow healthcare providers to use the vaccine by intradermal injection [administered between layers of skin] for individuals 18 years of age and older who are determined to be at high risk for monkeypox infection.
JYNNEOS has been tested in individuals with immunocompromising conditions and has been found to be safe and effective in the trials that were performed to support approval. It was initially developed specifically as an alternative for use in immunocompromised individuals in the event of a smallpox outbreak.
On the basis of the determination by the Secretary of the Department of Health and Human Services on Aug. 9, 2022, that there is a public health emergency, or the significant potential for a public health emergency, that has a significant potential to affect national security or the health and security of United States citizens living abroad, and the declaration on Aug. 9, 2022, that circumstances exist justifying the emergency use of vaccines, the FDA may issue an EUA to allow emergency use of unapproved vaccines or unapproved uses of approved vaccines.